Beyond diamonds: Search is on for rare carbon crystals
More than 140 unknown, carbon-based minerals are likely awaiting discovery

Scientists and amateur collectors search the ends of the earth for new minerals. Here, geochemist Daniel Hummer looks through the aluminum-rich waters at a site in northeastern Pennsylvania.
D. Hummer/Deep Carbon/Flickr
By Sid Perkins
Long ago, in Russia’s distant Northwest, a bird pooped. But this was not just any old splat of excrement. The feces landed on a very hot rock. It lay atop an underground coal fire. The intense heat quick-baked the poop. And that caused one of the substances previously dissolved in the feces to crystallize. With that, a new mineral was born.
Minerals are substances with a distinct chemical recipe. And the atoms of each mineral will always arrange themselves exactly the same way.
You’d expect to find crystals in rocks. You’d expect to find them in caves and mines. You would not expect to find one in baked bird poop.

Then again, few geologists would think to dig through bird feces. But those who dug through the Russian poop found something special. Unique even. It was a never-before-described type of crystal. The scientists named this new mineral tinnunculite (Tih-NUN-kew-lyte). That name comes, in part, from the scientific name of the European kestrel — Falco tinnunculus. It was this bird’s wastes that had rained down upon the hot Russian rock.
Tinnunculite is unusual in another way, too. It contains the element carbon.
What makes carbon minerals particularly interesting? For one thing, they’re somewhat rare. Only 8 percent of minerals contain this element; that’s 416 of all minerals known. Perhaps more importantly, carbon is a key element for life on Earth. In fact, many of the yet-to-be-discovered carbon-bearing minerals — just like tinnunculite — might be made either by living things or from compounds that they created.
There might be hundreds of undiscovered minerals on Earth, recent studies have suggested. Dozens — like diamonds — might contain carbon, geologists suspect. Now, the hunt is on for hidden relatives of the diamond. Some may be distant geological cousins. Others may seem even less related, like a nephew by marriage. Many scientists think carbon should be more heavily represented among the mineral-family tree. So they’ve begun a Carbon Mineral Challenge.
They launched it about 18 months ago. To date, scientists have already announced the discovery of eight new members of the carbon-mineral family. But there’s still plenty of time for ambitious rock hounds to make their mark on mineralogy before the search officially ends in late 2019.
Finding a new mineral means the discoverer gets to name their find. And they’ll get more than the chance to immortalize themselves in the pages of geology books. After all, they will be helping tell the story of our planet — both past and present. First, though, they’ll need to know where to search.
Fortunately, scientists have some good ideas on where to look.
Story continues below graph.
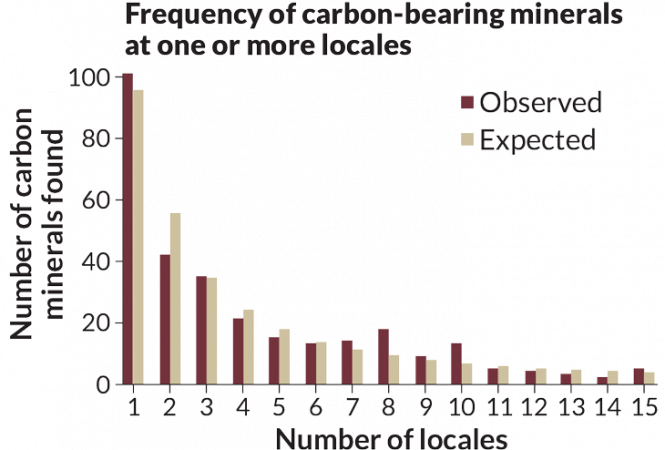
A census of minerals
Not every mineral on Earth has been discovered. Earth is, after all, a vast place, full of unexplored reaches. How many minerals might there be? Rare or everywhere, each has its own mix of elements. More than 5,000 minerals have been identified. It is possible that only a few of them have escaped notice. Or it could be most of them. It is probably impossible to know which is true. But to understand the complexity of the issue, it may help to know that the same rocks do not typically occur everywhere.
Yes, a few minerals are (forgive the phrase) as common as dirt. Perhaps surprisingly, however, most minerals are very rare. Fewer than one in every 500 have been found at more than 1,000 sites across the planet. More than half of the world’s minerals have been found at only five or fewer locations. And at least 1,000 minerals have been reported from only a single spot on Earth, notes Robert Hazen. He’s a geophysicist at the Carnegie Institution for Science in Washington, D.C.

So if one were on the hunt for crystals in the diamond’s family, where would one look? Hazen and his colleagues have a general idea. The group used statistics to analyze a large database of known minerals. The researchers noted how many sites host the common mineral. They compared these types of sites to the ones where rare minerals have been reported. This new analysis suggests that there should be at least 1,500 minerals awaiting discovery. Of them, about 140 should contain carbon, a second analysis concludes.
Minerals help tell the story of our planet’s water and carbon cycles. A mineral’s presence, for example, might help scientists puzzle out the precise conditions that existed when and where a crystal formed. (That’s because most minerals seem to form only under very particular conditions.) Plus, because minerals can continue to exist only under certain conditions, their presence can identify the local conditions that have existed since those minerals first crystallized. (One example: those coal fires underneath the site of that fried kestrel poop.)
The search is on
Anyone can participate in the Carbon Mineral Challenge. Amateur rock hounds are every bit as welcome as professional geologists, says Daniel Hummer. He’s a geochemist at Southern Illinois University in Carbondale. He’s also the scientist in charge of the search.

Finding a mineral is only the start of the challenge, though. Anyone who thinks she’s discovered a new one must prove it. She or her team must now collect a lot of data and answer many questions. Among them: What chemical elements are in the mineral’s recipe? How are the crystal’s atoms arranged? What does the mineral look like? What color is it? Is it transparent? How hard is it? Does it glow when certain colors of light shine on it?
The discovery teams must then send all of this information to the International Mineralogical Association (IMA). One of its special committees will then review the application. They must compare the supposedly new crystal to all known minerals to confirm whether it is unique.
IMA recognizes a few dozen new minerals each year, says Hans-Peter Schertl. He’s a mineralogist at Ruhr University in Bochum, Germany. He’s also an IMA officer. Sometimes approval is quick and easy. If the committee needs additional data, a decision might drag out for months or longer. That may reflect a need for more testing.
One strict requirement is that a sample be natural. It can’t have been made in a lab. (Researchers can make minerals by squeezing a rock under intense pressure, for example. But those minerals are not counted as new.) The sample also can’t have been made by other sorts of human interference. So, any unusual crystals that grow on the surfaces of rocks pulled from a mine and then dumped nearby wouldn’t qualify as new, says Schertl: “Those would just be pretty crystals.”
Story continues below video.
Where should we look?
Minerals can be rare for several reasons. They may form or remain stable only under a certain unusual mix of temperatures and pressures. They might include chemical elements that are rare to begin with and even rarer in combination. Or a mineral might form but almost immediately disappear. For example, some minerals are very hygroscopic. That means they pull moisture from the air, prompting themselves to dissolve. Finally, some minerals form in environments so remote or harsh that scientists hardly ever get near them. (Think deep-sea hydrothermal vents or active volcanoes.)
Some minerals present more than one of these challenges. Consider fingerite. Its chemical formula is Cu11O2(VO4)6. It is unstable under normal conditions, so its atoms don’t stay in their typical arrangement. Containing the rare pairing of copper and vanadium, it has been found in only one place: in Central America near the heat-belching fissures and holes atop El Salvador’s Izalco volcano.
Researchers looking for new carbon minerals could look for them anywhere. But some places are more likely to yield treasures. This means that if someone wants to find a new carbon mineral, they don’t have to search the entire planet. They can let science guide them.
For example, 14 different sites worldwide have already yielded 20 or more carbon minerals. Those sites include mines and quarries where people have been digging up rocks for years. Scientists could revisit those places, Hazen notes, looking for what might have been missed.
Or, they might simply take a closer look at rocks collected from there and now stored in some place such as a museum. Those samples might look like known minerals — but in fact have a different chemical recipe, says Hummer. Perhaps the mineral contains magnesium atoms instead of calcium. Or maybe it has sodium instead of potassium.
“Existing minerals that have a very similar formula can, in many cases, serve as a guide for what missing minerals might look like,” Hummer notes. In fact, similarities could be so strong that a new mineral might easily be overlooked. That means “it’s possible,” he says, “that some of these minerals are hiding in plain sight.”
Another search strategy: Go looking where a lot of other minerals have been found, not just carbon minerals. One example is the now-abandoned Eureka mine in northern Spain. In the past, miners brought up a lot of valuable uranium minerals from this site.
Story continues below interactive.
Tap the map to explore carbon mineral “hot spots” around the world.
Sites that have already proven rich in carbon-bearing minerals could be harboring more. Scientists and rock hounds aspiring to geologic fame should visit these locales (or analyze samples already collected there).
Source: D. Hummer/Southern Illinois Univ.
Like in many mines, water drips down Eureka’s walls. That water carries dissolved substances. They will be picked up by rocks as the fluid seeps downward through the rocks overhead. When the water evaporates in the tunnel’s cool air, some of the dissolved elements may combine to form new substances in solid form.
“The mine is a crystallization factory of weird minerals,” observes Jordi Ibañez-Insa. He’s a physicist in Spain at the Institute of Earth Sciences Jaime Almera in Barcelona.

To date, scientists have turned up 61 different minerals in just this one mine. The latest find is a carbon mineral called abellaite. It grows in small pincushions of tiny, needle-shaped crystals. Discovered in July 2010 (and approved by IMA in December 2015), the mineral has been found only along one 3-meter (10-foot) stretch of the mine’s tunnel, says Ibañez-Insa. He’s part of the team that submitted information about abellaite to the IMA. And he plans to spend more time at that mine in the future, looking for more undiscovered treasures. “I’m pretty sure we’ll find some more new minerals there,” he says.
And if looking in museums, mines, caves and other remote locations doesn’t yield new minerals, scientists can always go back to digging through bird poop. It’s paid off before.