First drug is found to block the new coronavirus
Early results suggest the drug, called remdesivir, speeds recovery from COVID-19
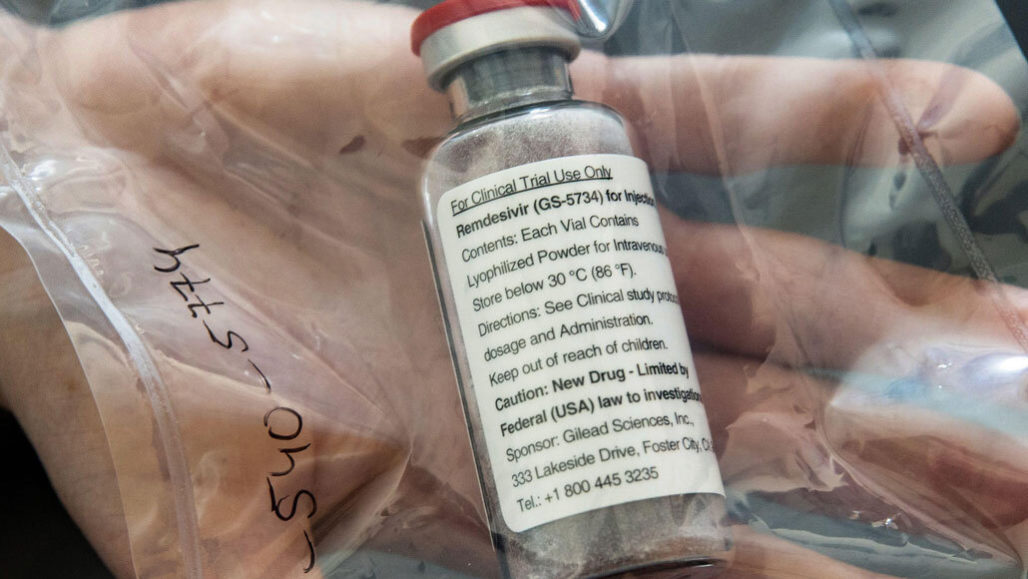
Remdesivir, an antiviral drug that stops some viruses from making copies of their genetic material, may help COVID-19 patients recover faster.
dpa picture alliance/Alamy Stock Photo
Early results from a new international COVID-19 trial in people “has proven that a drug can block this virus.” Anthony Fauci made the announcement April 29 at a White House news briefing. Fauci heads the National Institute of Allergy and Infectious Diseases in Bethesda, Md., which sponsored the trial.
The drug is called remdesivir (Rem-DESS-ih-veer). And it seems to speed recovery from the new coronavirus by 31 percent. What that means: Patients on the drug tended to be released from a hospital or were well enough to resume normal activity four days earlier than people who got a placebo. A placebo is pill or liquid that resembles a drug but contains no active ingredients. Sometimes people call placebos “sugar pills.”
“A 31 percent improvement doesn’t seem like a knockout 100 percent,” admitted Anthony Fauci. Still, he noted, “it is a very important proof of concept.”
The trial compared this drug to a placebo. Patients were randomly assigned to get either the drug or the placebo by an intravenous infusion. Neither the patients nor their doctors knew who was getting the real drug or the fake. Such randomized, double-blind, placebo-controlled trials are the gold standard for testing new drugs.
The trial began February 21 and involved 1,063 people hospitalized with COVID-19. The median time to recovery was 11 days for the remdesivir group. For those getting the placebo, it was 15 days.
Normally, researchers would not have announced the findings until the results had been reviewed by other scientists. But here the team chose to make the announcement early, Fauci said. “Whenever you have clear-cut evidence that a drug works, you have an ethical obligation to immediately let the people who are in the placebo group know so that they can have access [to the drug].”
Remdesivir will now be the standard of care against which all other drugs are judged, Fauci said.
Going forward, he noted, the initial trial will change. It will now compare this drug alone to its use with an antibody — one that aims to limit inflammation. Other drugs or antibodies may also tested to see if they do a better job than remdesivir.
Gilead Sciences of Foster City, Calif., developed remdesivir. The drug mimics a building block of RNA. That’s the coronavirus’s genetic material. When an infected cell attempts to copy the virus, the drug gets used instead of the usual RNA components. This cuts down how much virus is made.
Educators and Parents, Sign Up for The Cheat Sheet
Weekly updates to help you use Science News Explores in the learning environment
Thank you for signing up!
There was a problem signing you up.
Why remdesivir was tried
In petri dishes and animals, remdesivir had already been shown to be active in fighting a wide variety of RNA-based viruses. These included the coronaviruses that cause MERS and SARS. “It’s passed every single milestone. It works against every coronavirus we’ve tested,” says Mark Denison. He’s a virologist who was not involved in the study. Denison works at Vanderbilt University Medical Center in Nashville, Tenn.
In animals, the drug worked best when given early in infections, Denison says. It stopped or slowed the ability of the virus to reproduce. What the drug does not do, he notes, is block the body’s overactive immune response. When it goes into overdrive, the immune system can cause additional damage. And this has been seen in many severely ill COVID-19 patients.
Denison likens the new drug to a fire extinguisher. “If there’s a fire, and you put it out with the fire extinguisher, you’re not going to get burned.” But if you fall in the fire you’ll burn your arm. Using the fire extinguisher now may stop you from getting burned more, but it won’t heal the burns you already have.
Because such a drug is sent directly into the bloodstream, it must be administered by trained medical professionals. That makes remdesivir hard to give to people at home. But if you could do that early enough in the infection, Denison says, people might never become ill enough to need to go to the hospital. “You [could] convert this from being a lethal disease, to being a manageable, survivable disease,” he says.
And if such a drug could be given by mouth, it might even be used to prevent infections, Denison says.
Results from three other trials of the drug
Also on April 29, Gilead announced findings from another trial. It compared a five-day treatment of its drug against 10 days of the antiviral treatment. Here everyone got the drug. (That means there was no placebo for comparison). Of the 200 people who were treated for five days, 129 went home from the hospital by day 14. Of the 197 getting the longer treatment, 106 were discharged by day 14. Those results indicate that five days may be as good as 10 days for treating the infection.
“The study demonstrates the potential for some patients to be treated with a 5-day regimen,” the company said in a news release. That could greatly expand how many patients who could be treated with its current supply of the drug, it added. “This is particularly important in the setting of a pandemic, to help hospitals and health care workers treat more patients in urgent need of care.”
Treating earlier also proved better. Among patients who got treatment within 10 days of when symptoms first started, almost two-thirds (62 percent) were able to go home after two weeks in the hospital. Of those who got treatment later in the infection, slightly fewer than half (49 percent) were discharged after two weeks in the hospital.
A third and smaller, incomplete study was also published April 29. Its findings did not match those of the NIAID study. This placebo-controlled trial took place in 10 hospitals in Wuhan, China. That’s the pandemic first started. It found no clear improvement in recovery time among severely ill COVID-19 patients getting remdesivir.
Here, the median time to recovery for patients taking remdesivir was 21 days. For those getting the fake drug, it was 23 days. There was a trend suggesting remdesivir might speed recovery. But this was only in people who began treatment less than 10 days after their symptoms first showed up. Researchers in China reported their findings online in The Lancet.
Their trial was never fully completed. The reason: Wuhan’s lockdown effectively stopped transmission of the virus. The researchers could never fill the trial’s remaining slots. As a result, this study lacked the mathematical power to detect differences between the groups, Denison explains.
Finally, an international team of researchers gave remdesivir to 53 patients for “compassionate use.” These individuals had no access to participate in a clinical trial. All got at least one dose by March 7. Among these people, 36 needed less supplemental oxygen afterward. These findings were reported April 10 in the New England Journal of Medicine.