Immune targeting of cancers wins two a 2018 Nobel Prize
New therapies based on their ideas focus body’s immune cells against cancers

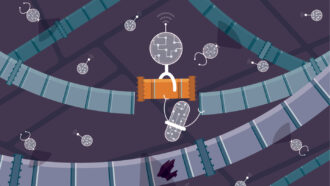
Rev up that immune system! Using the metaphor of a car, the green structures here illustrate how antibodies can take the brake off of the body’s immune system. This lets the body naturally attack cancer cells. The discoveries that make this therapy possible are behind the 2018 Nobel Prize for physiology or medicine.
The Nobel Committee for Physiology or Medicine. Illustrator: Mattias Karlén
By Tina Hesman Saey and Aimee Cunningham
Doctors used to target cancers only with scalpels, toxic chemicals and radiation. Recently, a new therapy has been emerging. It instead allows the body’s immune system to take out cancer cells. Two men who did the pivotal work that made such an immune therapy for cancer possible were today awarded the Nobel Prize in physiology or medicine.
James P. Allison, 70, works at the MD Anderson Cancer Center in Houston, Texas. He will share this year’s award with Tasuku Honjo, 76, of Kyoto University in Japan. At a ceremony in December, the two will equally share the prize of 9 million kronor. That’s equal to a little more than $1 million.
Discoveries by these men “have added a new pillar in cancer therapy,” says Klas Kärre. This immunologist works at the Karolinska Institute in Stockholm, Sweden. He also is a member of the Nobel committee that awarded today’s prize. Immune therapy against cancer is “a new principle,” he observes. Other therapies — such as surgery, radiation and chemotherapy — targeted tumor cells. The new strategy instead revs up a patient’s own immune system. It gives cellular players in that immune system permission to attack the cancer.
The basic discoveries by Allison and Honjo represent a new “landmark in the fight against cancer,” Kärre said as he announced today’s award.
CTLA-4 is the name of a protein on the surface of immune cells, known as T cells. Allison discovered that this protein holds T cells back from attacking cancer cells. You might think of it as acting like the brake on a car. Allison’s lab worked to release that brake. To do it, they developed an antibody against the protein. And that, his team showed, allows T cells to kill tumor cells.
In a series of experiments, Allison and his colleagues used this immune therapy in mice with cancer. The treatment actually cured the rodents or shrank their tumors.
The technique has worked especially well against a type of human skin cancer known as melanoma. In 2011, the U.S. Food and Drug Administration, or FDA, approved a drug to treat that type of cancer. Known as ipilimumab (Ih-pih-LIH-myoo-mab), it is sold under the brand name Yervoy. More recently, it also has been approved to treat colorectal cancer and a type of kidney cancer known as renal (REE-nul) cell carcinoma (Kar-sih-NO-muh).
But this therapy can sometimes have bad side effects. In some people, taking the brake off of the T cells allowed them to inappropriately attack healthy tissues. What’s more, some cancers don’t respond to this kind of immune brake-release therapy. Among them, notes Allison, are pancreatic cancer and glioblastoma. That second one is a type of brain cancer.
Honjo discovered the body also produces a second type of natural brake on T-cell action. It’s known as PD-1. And using antibodies to knock out this brake seems to work even better than Yervoy against cancer cells that have begun to spread throughout the body. This spreading disease is known as metastasis (Meh-TAS-tuh-sis). Until scientists learned how to take the PD-1 brake off of T cells, this spreading cancer could not be cured. In 2012, the FDA approved the first antibody to release the PD-1 brake on T cells.
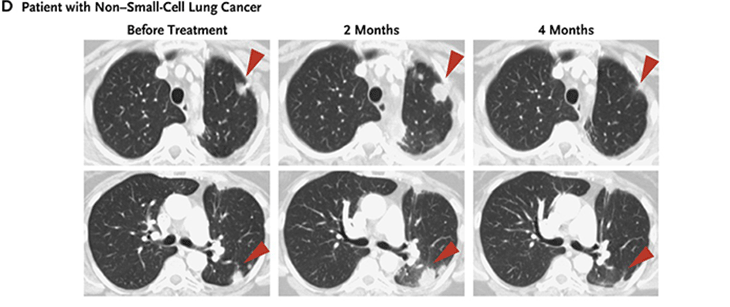
Now there are several antibodies against both PD-1 and its partner protein PD-L1. FDA has approved their use against melanoma, non-small cell lung cancer, kidney cancer, bladder cancer, head and neck cancers and Hodgkin lymphoma . Like the drugs that release the CTLA-4 brake, PD-1 antibodies have side effects. Still, they are generally milder than those due to the CTLA-4 brake release.
Reacting to the Nobel announcement
Norman “Ned” Sharpless directs the National Cancer Institute in Bethesda, Md. The new brake-release drugs are known as “immune checkpoint” therapies. And they have been a boon for cancer patients, he says.
“We’re not curing everybody,” he notes. “But in some cancers, 20 to 30 percent of patients will have substantial benefit.” Before, he notes, “We had nothing for those people.”

Physiology (Fiz-ee-OL-oh-gee) is the science of how the body works. Medicine is the field of fighting disease. Beyond their work in cancer, Allison and Honjo have contributed greatly to physiology, says Sharpless. In this case, they helped provide a better understanding of how the immune system works. The Nobel selection committee chose to honor the men’s work in cancer treatment, or medicine. However, he argues, they “also deserve lifetime achievement awards for their contributions to science.”
“I did not get into these studies to try to cure cancer,” he explained. “I got in to them because I wanted to know how T cells worked.” That type of work is known as basic research. Other approaches to treating cancer, such as with vaccines, hadn’t been as successful. Perhaps, Allison now says, that’s because “people started with insufficient knowledge.”
Honjo also pointed to the value of basic research here. PD-1’s discovery in 1992 “was purely a matter of basic scientific research,” Honjo said at a news conference today. But then it led to actual treatments. In time, he notes, his patients began to tell him: “This treatment has improved my condition and given me strength again. And it is all thanks to you.” With such comments, Honjo says, “I really began to understand the meaning of what my work had accomplished.”
Allison learned of the prize from his son. He had called his dad in his hotel room in New York City where Allison is attending a conference on cancer research. Soon, friends were calling and coming to Allison’s hotel room to celebrate. “We had a little party in the room this morning,” he said during a news conference.
Allison gave a shout-out to cancer patients. “We’re making progress,” he said. He wants to increase the number of them who can be helped by immunotherapy. “We know how to do it, we’ve just go to learn to do it better.” And one way may be to use it along with other therapies, such as radiation or chemotherapy.
Allison and Honjo will receive a medal and their prize money at a December 10 awards ceremony in Stockholm.