Common drug, dexamethasone, appears first to cut COVID-19 deaths
It might save one in every eight people on ventilators and one in every 25 on oxygen
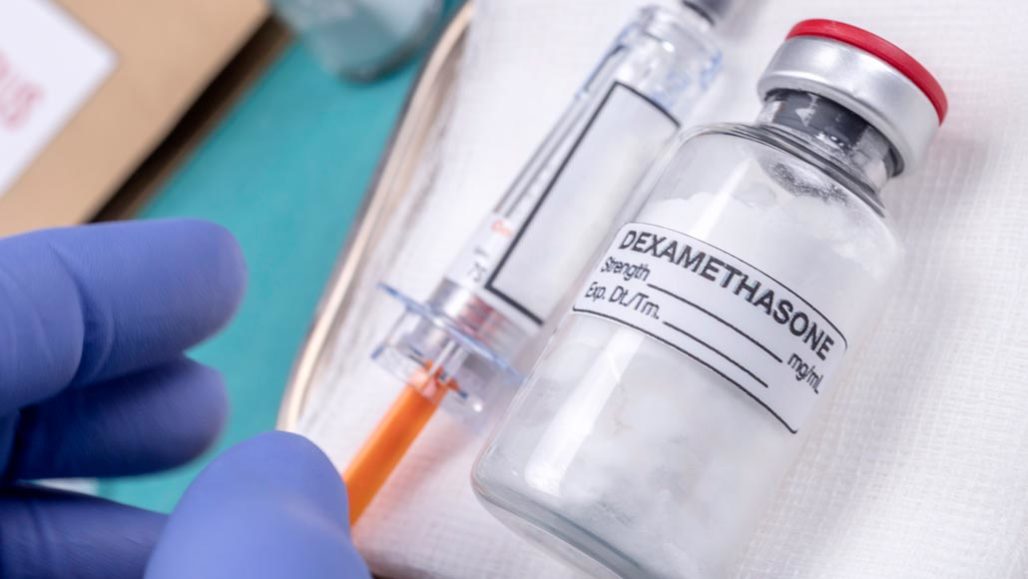
This commonly used, decades-old drug reduced the number of deaths among COVID-19 patients on ventilators. Scientists reported the findings in mid-June.
Digicomphoto/iStock/Getty Images Plus
A widely available and low-cost drug may save the lives of some patients hospitalized with COVID-19. The finding comes from preliminary data collected as part of a large clinical trial. The drug helped seriously ill patients who were on ventilators or getting extra oxygen.
The drug is dexamethasone (Dex-uh-METH-ah-sown). It’s a type of synthetic steroid hormone. Doctors have prescribed it for decades to fight inflammation. Here, it was tested on COVID-19 patients at more than 175 hospitals in the United Kingdom.
Compared to standard care, use of this drug cut the death rate by about one-third in people on ventilators. (Those machines effectively breathe for the patients.) It cut death rates by one-fifth in patients who could breathe on their own but needed some supplemental oxygen. There appeared to be no benefit for coronavirus patients who didn’t need extra oxygen.
Last month, scientists reported promising data on a different drug, which appears to shorten recovery time for seriously ill patients.
The findings for dexamethasone have not yet appeared in a scientific journal. They were instead issued in a June 16 news release. This means outside scientists have not yet had a chance to review all the data.
Once they do and the results still are able to hold up to scrutiny, this drug would be the first shown to cut the risk of death from COVID-19. When it comes to the coronavirus, says Martin Landray, “This is the first drug that says, yes, we can increase your chances of survival.” Landray is a heart specialist in England at the University of Oxford.
Educators and Parents, Sign Up for The Cheat Sheet
Weekly updates to help you use Science News Explores in the learning environment
Thank you for signing up!
There was a problem signing you up.
Equal parts excitement and caution
In the new study, 2,104 patients got 6 milligrams of dexamethasone once a day for 10 days. Another 4,321 people did not get this drug. The study was stopped early once a review committee felt enough patients had been enrolled to identify whether the drug worked. Landray and his colleagues found that taking the drug could prevent one death for every eight patients on ventilation, and one death for every 25 patients needing extra oxygen.
“It’s not a fix. It’s not a cure. It’s not a miracle,” says Landray. “But it is really, really useful.” He expects doctors around the world will quickly look to use the drug for COVID-19 patients who have trouble breathing.
Rajesh Gandhi is an infectious diseases doctor in Boston at Massachusetts General Hospital and Harvard Medical School. “These are potentially very exciting data,” he says. Gandhi helped write guidelines for COVID-19 treatment issued by the National Institutes of Health and by the Infectious Diseases Society of America. Still, he will wait until he sees the full report to decide whether the results warrant changing treatment of severely ill patients. “Right now we have very limited information. But if it’s borne out, it could be really exciting.”
The new results show the drug could be important for the sickest patients with COVID-19. However, he points out, those are only one in every 20 patients diagnosed with the coronavirus.
For most patients, then, the drug likely would not do any good. It might even cause them harm. Like other steroids, this drug tamps down the immune system’s response to invading organisms. As such, it has worsened some viral infections, such as SARS and the flu. Because SARS is a coronavirus, researchers had thought the drug might also worsen recovery from COVID-19.
The way the results were released has some scientists worried that they may not hold up. Landray says he understands that criticism. However, he explains, with so many people suffering from the pandemic, his team felt it was important to “get the results out” so that the “world at large can see them and make their decisions.”
He and his colleagues are now writing up a scientific paper with lots of details. They know it will take time for their study to be reviewed and published. Meanwhile, Landray says, this commonly prescribed drug “is already sitting in hospitals now. It doesn’t cost much money and the benefits are really obvious.”
Brian Garibaldi is hopeful that the results will hold up. Anything that could cut the virus’ death rate “would be a game changer,” says this doctor at Johns Hopkins Medicine in Baltimore, Md. But, he adds, doctors are unlikely to change how they treat severely ill COVID-19 patients based on the limited data presented last week. He says that he won’t use the drug for his patients until he sees all the data.