
Chemistry
Pollution power? A new device turns carbon dioxide into fuel
Scientists made a device that converts the greenhouse gas into formate. This salt can then run a fuel cell to make electricity.
By Laura Allen
Come explore with us!

Scientists made a device that converts the greenhouse gas into formate. This salt can then run a fuel cell to make electricity.

How does a parachute work? Do bigger parachutes work better than smaller parachutes? Find out in this science project whether the size of the parachute matters.

Neutrons are one of the main building blocks of atoms and have no electric charge.
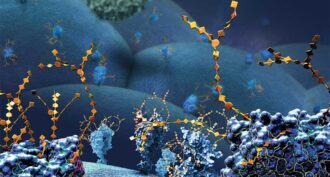
This so-called ‘click chemistry’ allows scientists to build complex molecules in the lab and in living cells.

Three pioneers in quantum physics share the 2022 Nobel Prize in physics.

These positively charged particles are important building blocks in atoms.

Ultraviolet light, sulfite and iodide break down these PFAS molecules faster and more thoroughly than other methods.

Scientists 3-D printed the new fabric, which has even more tricks up its sleeve — such as conducting electricity and resisting radio waves.

Researchers have developed a liquid metal that breaks down carbon dioxide in the air, converting it from a climate threat into a valuable raw material.

Data show a major class of long-used “eco-friendly” copper chemicals unexpectedly react with soil, making gases harmful to Earth’s protective ozone layer.