Nobel goes for making white LEDs possible
The three recipients developed blue LEDs, a necessary and missing constituent in creating white light with LED technology

The technology that allows today’s smartphones, TVs and more to produce a white or blue light was challenging to develop. Three pioneers who made it happen received the 2014 Nobel Prize for physics.
Tomwang112/ iStockphoto
By Janet Raloff
That bluish-white light glowing from the screens of most new televisions, smartphones, laptops and tablet computers? It also comes from light-emitting diodes, better known as LEDs. Many businesses light their work spaces with LEDs. More and more, LEDs light outdoor street signs and traffic lights. Some homeowners have begun turning to this new form of digital — or solid-state — lighting to illuminate their rooms. And most cars and trucks now use these same LEDs in their tail lights. Three scientists have now won the 2014 Nobel Prize in physics for developing the technology that has made this lighting possible.
The physicists did not win for making the white LEDs that have become so common. That invention would come later. Their achievement was creating a building block of white LEDs 20 years ago: the blue LED.

To create white light using LEDs, a device must combine at least three different wavelengths (colors). Engineers typically mix the three primary colors: red, green and blue. The need to do this mixing is old news, observes Per Delsing. This has been “known since Isaac Newton in 1671,” he says. A physicist at Chalmers University of Technology in Göteborg, Sweden, Delsing chaired this year’s Nobel Committee for Physics.
Although red and green LEDs have existed for about 50 years, back in the late 1980s, a blue LED was still only a dream. Many companies set teams of their researchers to work creating one. They all failed, Delsing says. But in Japan, three scientists kept trying. Eventually, he notes, their efforts paid off.
On October 7, the Royal Swedish Academy of Sciences in Stockholm named them winners of 2014 Nobel Prize in physics. Isamu Akasaki and Hiroshi Amano of Nagoya University in Japan and Shuji Nakamura, who now works at the University of California, Santa Barbara, will split the roughly $1.1 million award. Nakamura made his early contributions to LEDs while at Nichia Chemicals in Tokushima, Japan.
At least one-fifth of all energy used throughout the world powers lighting. LEDs convert up to 50 percent of electrical energy entering them into light. The most recent record is an LED that converts one watt of power into slightly more than 300 lumens of light. That’s much better than the 70 lumens per watt achieved by fluorescent lights. And it far surpasses the paltry 15 lumens per watt offered by those old-school incandescent light bulbs. That’s one reason the sale of incandescent lights is being phased out in many parts of the world. Another reason: Other types of lights, especially LEDs, last much, much longer.
Chemist and inventor Alfred Nobel, who created dynamite, made a fortune developing weapons. After his death, some of that money was used to fund an account that now pays out the prizes that carry his name. “I really think that Alfred Nobel would be very happy about this prize,” says Delsing. Nobel would have appreciated, Delsing says, that these inventors created “something that will benefit most people.”
The science behind it all
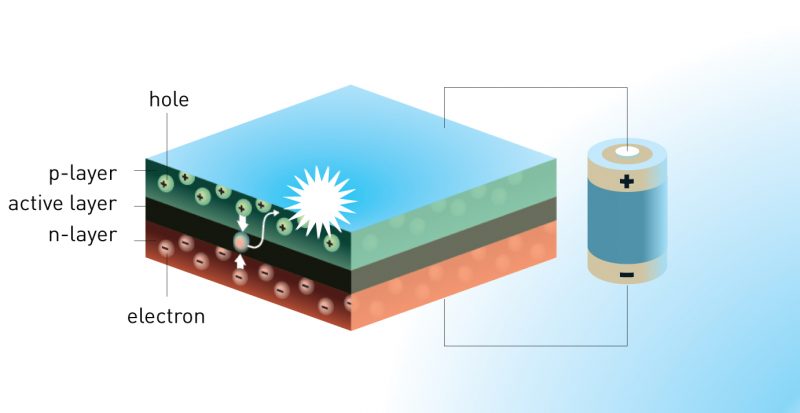
LEDs are small. Many devices are far smaller than a thumbnail. Individual components can be the size of a grain of sand. They are made of a sandwich of materials. One outside layer is known as a “p” layer. In the middle is an “active” layer. On the other side of it: a so-called “n” layer.
The n layer must have an excess of electrons. These are subatomic particles and the basis of an electric current. They possess a negative electric charge. The p-type layer must have an excess of holes. They’re the absence of electrons in a semiconductor material. Some physicists describe them as being like bubbles in a bottle of water, where those bubbles are the absence of water.
Applying an electrical voltage to LEDs causes electrons inside their receiving section to become highly energized. Some of these electrons then jump into the active layer of the device (see diagram above). At the same time, holes jump into it from the p-type layer. This process radiates light. The energy required for electrons to cross into the active layer determines what color an LED emits.
Delsing says his committee appreciated two particular achievements by this year’s winners. First, Akasaki and Amano succeeded in developing a good semiconductor material, the heart of any LED. For the new blue devices, this semiconductor is gallium nitride. “And that was very hard to grow,” Delsing says of the crystals. The scientists’ second major achievement was to dope it — insert into it — electrons and holes.
A particular challenge was creating the hole-enriched p-type layer. Akasaki and Amano succeeded in the late 1980s.
“In semiconductors like this, you dope both electrons and holes,” explains Delsing in a YouTube interview provided by the Nobel Committee. “And it was the hole doping which was very difficult. But [this year’s winners] succeeded.”
In 1988, Nakamura began working on blue LEDs and ultimately created the gallium nitride material through a different process. Within four years, he created a working p-type layer through a simpler and less expensive method than the one developed by Akasaki and Amano. Over the coming years, the three would improve blue LEDs even more. Those blue LEDs also would become the basis of a new blue laser. The short wavelength of its beam meant it could write more data onto storage devices than other types of light. This “quickly led to the development of Blu-ray discs with longer playback times, as well as better laser printers,” the Nobel Committee says.
Sweden’s King Carl XVI Gustaf will give Akasaki, Amano and Nakamura each man a medal and a document confirming his prize award at a Dec. 10 ceremony in Stockholm.
Power Words
digital (in electronics) A term for the type of technology based on semiconductors.
electron A negatively charged particle; the carrier of electricity within solids.
doping (in electronics) The deliberate insertion of something into a crystalline semiconductor material. For instance, manufacturers may dope a material with electrons or electron holes.
electronics Devices that are powered by electricity but whose properties are controlled by the semiconductors or other circuitry that channel or gate the movement of electric charges.
fluorescent lighting A technology that uses electricity to create glowing tubes of mercury vapor. They have been around since the late 1890s.
holes (in electronics) The absence of normally expected electrons in a semiconductor crystal.
incandescent lighting The old-style lighting technology that relied on a glass bulb. Electricity passing through the bulb heated a thread-like tungsten filament, making it glow white hot. Thomas Edison commercialized this technology in 1879. By that time, the technology was already about 50 years old. Incandescent lights used to illuminate everything from tiny flashlights to whole rooms. Many governments have moved to ban these bulbs because they waste so much of their energy as heat.
laser A device that generates an intense beam of coherent light of a single color. Lasers are used in drilling and cutting, alignment and guidance, in data storage and in surgery.
LED (light-emitting diode) A type of semiconductor device that produces light.
lumen (in lighting) Units that describe the visible light given off by a source, such as a light bulb. For instance, an old style, 100-watt incandescent light bulb typically emitted about 1,600 lumens when it was new.
n-type layer (in solid-state electronics) A layer of semiconductor material that has been doped — inserted with — electron holes.
p-type layer (in solid-state electronics) A layer of semiconductor material that has been doped — inserted with — bonus electrons.
semiconductor A material that sometimes conducts electricity. Semiconductors are important parts of computer chips and certain new electronic technologies, such as light-emitting diodes.
solid state A term for electronics technologies that create circuitry or devices from solid materials known as semiconductors. As they work, their electrons (or other charge carriers) remain confined entirely within the solid material.
voltage A force associated with an electric current that is measured in units known as volts. Power companies use high-voltage to move electric power over long distances.
watt A measure of the rate of energy use, flux (or flow) or production. It is equivalent to one joule per second. It describes the rate of energy converted from one form to another — or moved — per unit of time. For instance, a kilowatt is 1,000 watts, and household energy use is typically measured and quantified in terms of kilowatt-hours, or the number of kilowatts used per hour.
wavelength The distance between one peak and the next in a series of waves, or the distance between one trough and the next. Visible light — which, like all electromagnetic radiation, travels in waves — includes wavelengths between about 380 nanometers (violet) and about 740 nanometers (red). Radiation with wavelengths shorter than visible light includes gamma rays, X-rays and ultraviolet light. Longer-wavelength radiation includes infrared light, microwaves and radio waves.







