Chemistry Nobel honors pioneers of world’s smallest machines
These three chemists paved the way for molecular motors

This microscopic automobile is a “nanocar.” The creators of miniature machines such as this one have won the 2016 Nobel Prize in chemistry.
RANDY WIND/MARTIN ROELFS; NATURE
By Tina Hesman Saey and Thomas Sumner
The world’s smallest motors — ones far too small to see with the eye — may soon drive new innovations in chemistry, biology and computing. Three pioneers of such nanotechnology were honored with the 2016 Nobel Prize in chemistry.
The Nobel Committee in Stockholm, Sweden, announced the winners on October 5. Jean-Pierre Sauvage works at the University of Strasbourg, France. James Fraser Stoddart is at Northwestern University in Evanston, Ill. And Bernard Feringa works at the University of Groningen in the Netherlands.
“If you had to choose three people at the top of the field, that’s it. These are the men,” says James Tour. He’s a nanomachines chemist at Rice University in Houston, Texas. To his mind, “It is a well-warranted prize.” That means that the Nobel committee selected the right people.
Giving attention to the skyrocketing field of molecular motors (which this award will certainly do) should inspire more children to become scientists, says Donna Nelson. She’s an organic chemist at the University of Oklahoma in Norman. She’s also president of the American Chemical Society, based in Washington, D.C. This attention will likely also draw more funding for work in this area. As such, she says, this award “will benefit not only these three chemists, it will benefit the entire field of chemistry.”
Why create nano-bots?
Scientists have envisioned machines built on the scale of molecules since at least the 1960s. But reliably building their complex structures proved challenging. That began to change in 1983. That’s when Sauvage figured out how to loosely link ring-shaped molecules into a chain. These were known as catenanes (KAT-eh-nayns). These chains set the stage for the rest of the field.
(Story continues below image)
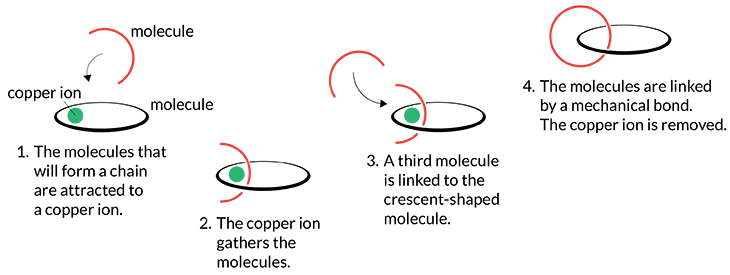
Stoddart improved the efficiency of making these chains. Before long, he could produce large quantities of them. By 1991, he had clipped such ring-shaped molecules to a central axle. That created what’s now known as a rotaxane. He and his colleagues learned to control how the rings slide along the axle. This formed a simple molecular switch. One day, such switches might be used to make molecular computers or new systems to deliver drugs to a desired site in the body.
Stoddart designed molecular “muscles” with interlocking ring molecules and axles. His group has since gone on to make molecular elevators and pumps, working with the same molecules.
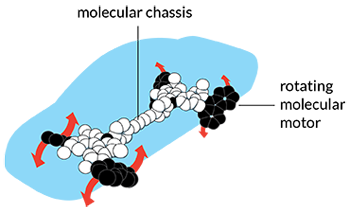
Feringa ramped things up another notch in 1999. That’s when he fashioned the first molecular motor. Things move so differently at the molecular scale that many researchers weren’t sure anyone could precisely control the motion of molecular motors, recalls Raymond Dean Astumian. He works at the University of Maine in Orono. But Feringa got it to work. His innovation was to devise asymmetric molecules that would spin in only one direction when hit with a pulse of light.
These were very tiny motors. Some 50,000 of them could fit side-by-side across the width of a human hair, notes Tour. Alone, one spinning motor wouldn’t do much. Harnessed together in large numbers, however, these motors could accomplish quite a lot, he says. Groups of the whirring devices, powered by light, can rotate a glass rod thousands of times their size. Feringa even harnessed his motors into a four-wheel-drive “nanocar.”
The promise of nano-machines
Over the decades, molecular machines have seen big improvements. And the work of the three new Nobel winners contributed a lot to that, says Rigoberto Advincula. He’s a chemist at Case Western Reserve University in Cleveland, Ohio.
Scientists today have a better understanding of how to create molecules that more reliably bend, loop and connect to form desired shapes. “You don’t have tweezers to put them together,” Advincula notes. Engineers have to design a template. Then they arrange to have a particular chemical reaction occur on that template. Depending on the reaction, it might make a thread go through some ring, for instance. That, he explains, might help the ends of two threads meet.
Ever newer techniques make it easier to create more challenging shapes. One day, scientists expect to be able to scale up the process. That could allow for the design of molecular machines for everything from energy harvesting to manufacturing complex chemicals.
But that’s still a long way off. Right now, no one really knows what particular sorts of useful machines chemists will put assemble from their molecular toolboxes.
Indeed, when people ask Feringa what his nano-motors might be used for, he says he “feels a bit like the Wright brothers.” Right after their first flight, these airmen would have had a hard time explaining why the world needed a flying machine. Feringa suspects there are “endless opportunities” for making good use of super miniscule motors. They might include nanomachines that seek and destroy tumor cells throughout the body, he says. Or maybe they will deliver drugs to just the cells that need them.
Stoddart applauded the Nobel committee for recognizing “a piece of chemistry that is extremely fundamental in its making and being.” Sauvage, in particular, created a new type of molecular bond in order to forge his chain, Stoddart noted during a press conference. “New chemical compounds are probably several thousand a day, worldwide. New chemical reactions, well, maybe a dozen or two a month.” But when it comes to creating new bonds, he noted, “they are few and far between. They are really the blue moons. So I think that’s what’s being recognized, more than anything.”
Sauvage, Stoddart and Feringa will receive their awards at a December 10 ceremony in Stockholm. They will share a prize of 8 million Swedish kronor (or about $931,000).
Their award is named for Alfred Nobel. Best known as the inventor of dynamite, he was a wealthy man when he died on December 10, 1896. In his will, Nobel left much of his fortune to create prizes to those who have done their best for humanity in the fields of physics, chemistry, physiology or medicine, literature and peace.
Francis Villatoro







