CRISPR enters its first human trials
The gene-editing technology will target cancer, blood disorders and blindness
Scientists are beginning human trials to treat disease with the “molecular scissors” known as CRISPR/Cas9. Some people with a form of inherited blindness will have it injected into their eyes, where researchers hope it will snip out a mutation.
TRAFFIC_ANALYZER/GETTY IMAGES PLUS
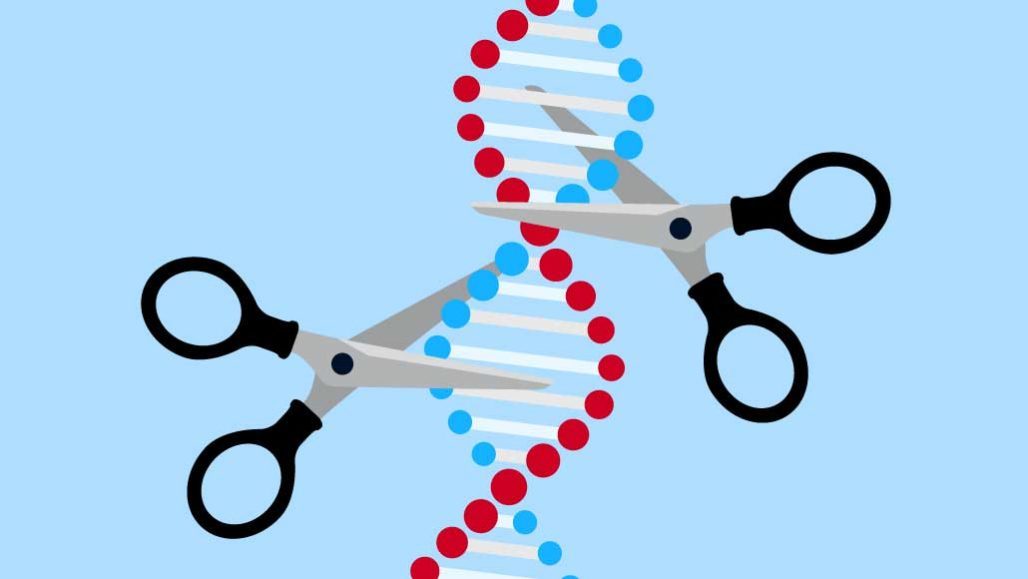
The answer to some genetic diseases may be a powerful “molecular scissors.” Known as CRISPR/Cas9, it has healed genetic diseases in some animals. Soon, doctors may learn how well it works in people. The first human studies to test its promise are just getting underway.
CRISPR/Cas9 cuts through DNA. But it doesn’t randomly chop. This gene editor comes in two parts. The CRISPR part is a short piece of genetic material called a guide RNA. Like seeing-eye dogs, these guides lead Cas9, an enzyme, to where it needs to go. Scientists can use CRISPR/Cas9 to intentionally break some genes or repair others.
In the first group of human trials, scientists are using the technique to fight cancer and blood disorders. Other researchers are set to study how CRISPR/Cas9 works inside the human body. In one upcoming trial, people with an inherited blindness will have the molecular scissors injected into their eyes.
If successful, such tests could lead to CRISPR trials for diseases affecting millions of people.
Out of body experience
In one pioneering trial, researchers at the University of Pennsylvania in Philadelphia are treating two people with recurring cancers. One has multiple myeloma (a disease that starts in the blood’s plasma cells). The other has a sarcoma. Both patients received a type of immune cell that had been programmed with CRISPR to go after cancer cells. (Similar trials are under way in China.)
Another trial is under way for two blood disorders: sickle-cell disease and beta-thalassemia (Bay-tuh Thah-lah-SEE-mee-uh). Both result from defects in the gene for hemoglobin. That’s the oxygen-carrying protein in red blood cells.
People who inherit a sickle-cell mutation or a beta-thalassemia mutation normally get these diseases, says David Altshuler. He’s the chief scientist at Vertex Pharmaceuticals. It’s a drug company based in Boston, Mass., and London, England. Changing a different gene may help people avoid these disorders.
In the womb, one form of hemoglobin helps fetuses grab extra oxygen from their mother’s blood. The body normally stops making this hemoglobin right after birth. But in some people, a harmless gene variant causes fetal hemoglobin to be produced throughout their lives.
People who have mutations that cause beta-thalassemia or sickle cell disease didn’t get either disorder if they also had the lifelong fetal-hemoglobin gene.
Vertex and CRISPR Therapeutics, a company in Cambridge, Mass., are now testing whether CRISPR/Cas9 can engineer changes in the body to mimic the change that keeps fetal hemoglobin turned on for life.
Both the cancer and blood-disorder trials take cells from people’s blood. DNA in those cells is edited with CRISPR/Cas9 in lab dishes. Scientists can test the cells to make sure the DNA has been edited properly. In the case of the blood disorders, CRISPR/Cas9 cuts a piece of DNA that usually helps turn off fetal hemoglobin after birth. Now the fetal-hemoglobin gene can turn on again. Doctors then return these edited cells into the sick people.
One person was given this treatment for beta-thalassemia in February. One got the treatment for sickle-cell disease in July.
Inside job
Other scientists are trying something much harder: editing DNA in cells that are still inside the body. Researchers will test this in people with an inherited type of blindness.
Some people have a mutant form of the gene known as CEP290. It causes rod cells in the eye’s retina to die, leading to blindness. In July, two companies started recruiting patients for a clinical trial of the gene editor.
One company is Editas Medicine of Cambridge, Mass. Editas is working with the drug maker Allergan. In their trial, CRISPR/Cas9 will make two cuts in the defective gene. This should snip out the troublesome piece of DNA.
The first people to get the test therapy will be adults who are nearly blind, says Charles Albright. He’s the chief scientist at Editas. “We’re going into arguably the most difficult patients to start with,” he says.
Small amounts of the CRISPR editor will be injected under the retina to test the treatment’s safety. Such low doses may not improve vision. But if those doses prove safe, later volunteers will get higher doses. The researchers may also test the therapy in children.
Editing as few as 10 percent of retinal cells might help restore some sight, Albright says. In animal tests, CRISPR edited up to about 60 percent of cells in mice and almost 28 percent in monkeys. The study’s authors reported their accomplishment in the February Nature Medicine.
Even if these first trials don’t work quite as hoped, CRISPR won’t be shelved, Albright says. “This is a technology that’s here to stay.”
“CRISPR is so intriguing,” adds Laurie Zoloth, “and so elegant.” A bioethicist, she works at the University of Chicago Divinity School in Illinois. Science, she says, just has to explore whether this new technology can live up to the hype.